Found some plots I took while curing. Every time I would turn the skimmer off then on, you can see the rise and then drop in pH.
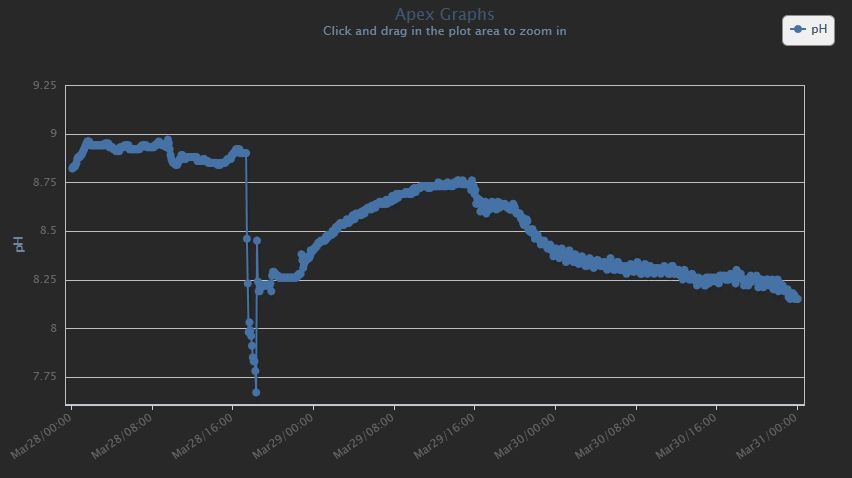
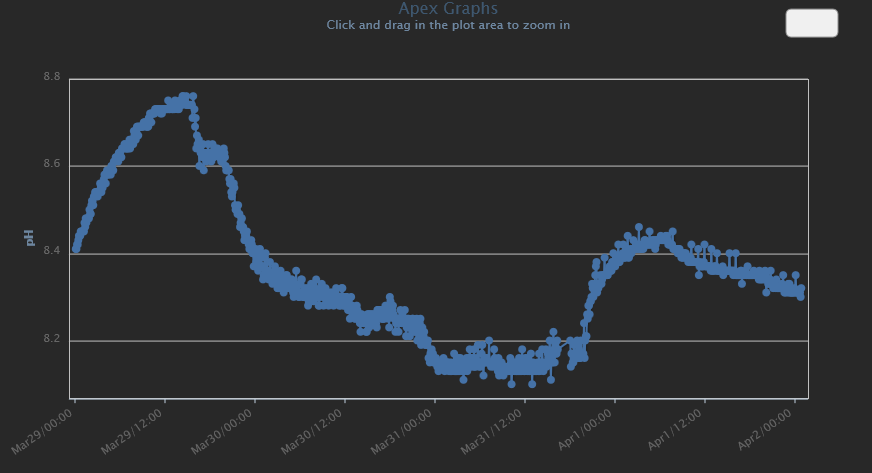
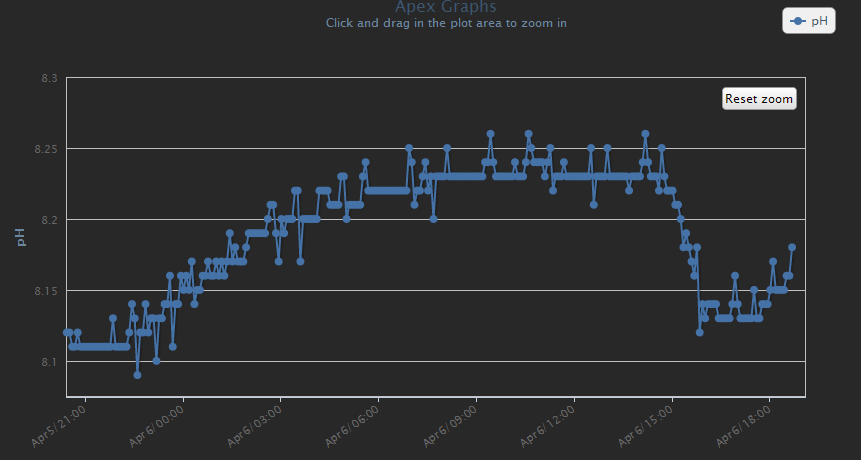


Unfortunately, I didn't capture the initial pH drop, but I think this shows what I'm talking about. Basically - the water and air will find equilibrium. If my pH is too low, I turn on my skimmer and up it goes. If my pH is too high, I turn on the skimmer and down it goes.
It doesn't matter if this is CO2, carbonate or bicarbonate... it is available and is pulled in from the air.
To keep Alk and Ca, I dose kalkwasser and that also impacts my pH but the primary driver is still CO2 in the air.
